E-Mail: lorotae@gmail.com
Dr. Lorena Lobos-González is a Biochemist from the Faculty of Chemistry of the Catholic University of Valparaíso, she obtained her Master’s degree (2006) and PhD in Biochemistry (2011) at the Faculty of Chemical and Pharmaceutical Sciences, University of Chile. She’s is a scientist whose skills allow her to work in areas such as teaching, research and biotechnology, among others.
On the other hand, Dr. Lobos-González is Associate Researcher in the Center for Chronic Diseases of the University of Chile (FONDAP- ACCDiS) and Assistant Professor in the Regenerative Medicine Center, Faculty of Medicine German Clinic, Universidad Del Desarrollo. At the beginning of the current year, she was incorporated as regular academic professor in the Institute of Biomedical Sciences (ICBM), Faculty of Medicine, University of Chile.
She has extensive research experience in the use of syngeneic immunosuppressed and immunocompetent murine models to evaluate gene pharmacotherapy success in cancer settings. Experimental approaches employed include resective surgeries, patient explants, PDX models and metastasis models. Dr. Lobos-González completed Phase 1A-1B trials, in collaboration with the University of California (UCLA), USA, developing a pharmacogenic type AS-ODNs targetting ncmtRNAs. On the other hand, with regards to teaching Dr. Lobos-González has a good command of didactic techniques, dynamics in learning and evaluation, skills she accured by training in pedagogy that does not lead to a degree.
Of rate is her ability to direct multidisciplinary teams to achieve a common goal. Recently, she complet a Startup Fondecyt Project on an innovative topic, in the area of cell communication via exosomes, involving at in vivo studies. Currently, Dr. Lobos-González leads a Regular Fondecyt on her current line of research, which is oriented towards the study of how microvesicles and exosomes derived from breast cancer tumor cell lines prepare metastatic niches in immunosuppressed models of breast cancer. In addition, she directs a Fondef I+D project that seeks to validate a protein present in these plasma microvesicles as a marker in patients with breast cancer, post-resective surgery. These studies are being carried out at the Center for Regenerative Medicine.
“Lactadherin in exosomes secreted by breast cancer cells interacts with αvβ3 integrin in epithelial cancer cells, leading to epithelial-mesenchymal transition and with αvβ3 integrin in endothelial cells, leading to increased endothelial permeability and metastasis post-surgery”.
Contribution: Principal Investigator, Regenerative Medicine Center, Faculty of Medicine, Universidad Del Desarrollo.
Period: April 2021 to April 2025.
Management: By own
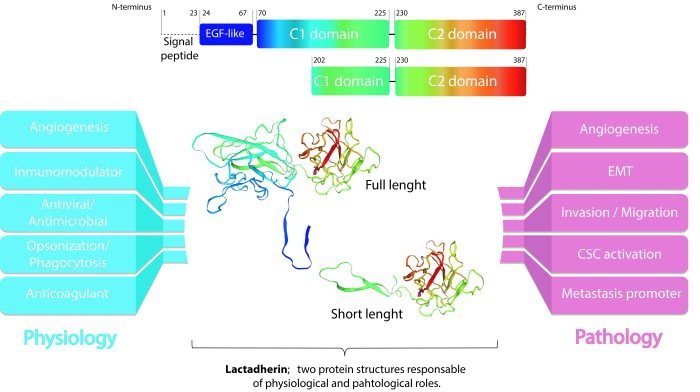
Figure 1: Lactadherin structure and possible roles in physiologic and pathologic states. From: Durán-Jara E, Vera-Tobar T and Lobos-González L. (2022).
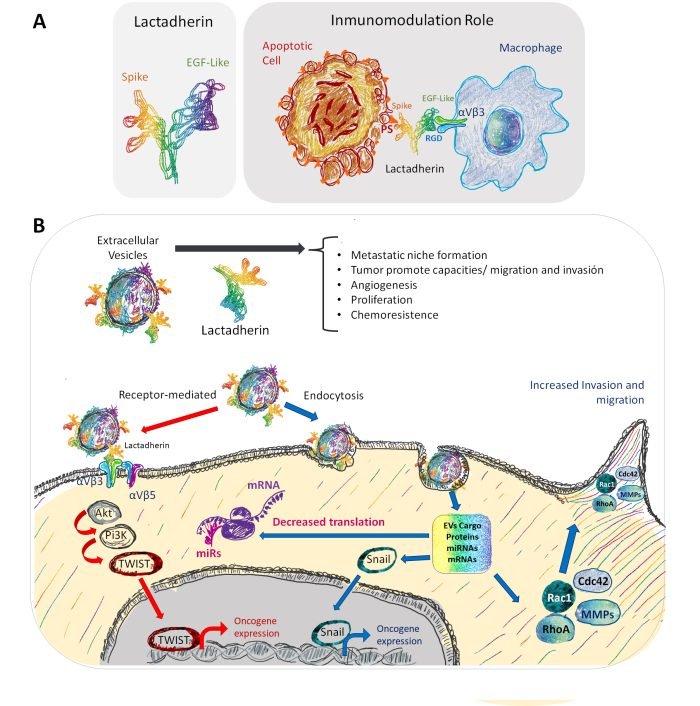
Figure 2 Representation of the immunomodulatory role and other possible roles of lactadherin present in EVs. (A) In non-lactating conditions, epithelial cells have low basal lactadherin expression. (B) However, in a tumor context, lactadherin overexpression is correlated with tumor progression through several ways, one of them being its presence in EVs. In this EV setting, lactadherin may be promoting tumor cell survival, migration, and invasion capacities, promoting angiogenesis and EMT. Also, lactadherin presence on EVs membranes could have a role as a targeting ligand for EVs towards recipient cells. From: Durán-Jara E, Vera-Tobar T and Lobos-González L. (2022).
“Validation of the Lactadherin marker present in plasma microvesicles as indicator of post-surgery recurrence/metastasis in breast cancer”.

Associated Institutions
Management: Director, Regenerative Medicine Center, Faculty of Medicine, Universidad Del Desarrollo.
Period: January 2022 to October 2024.
Management: By own
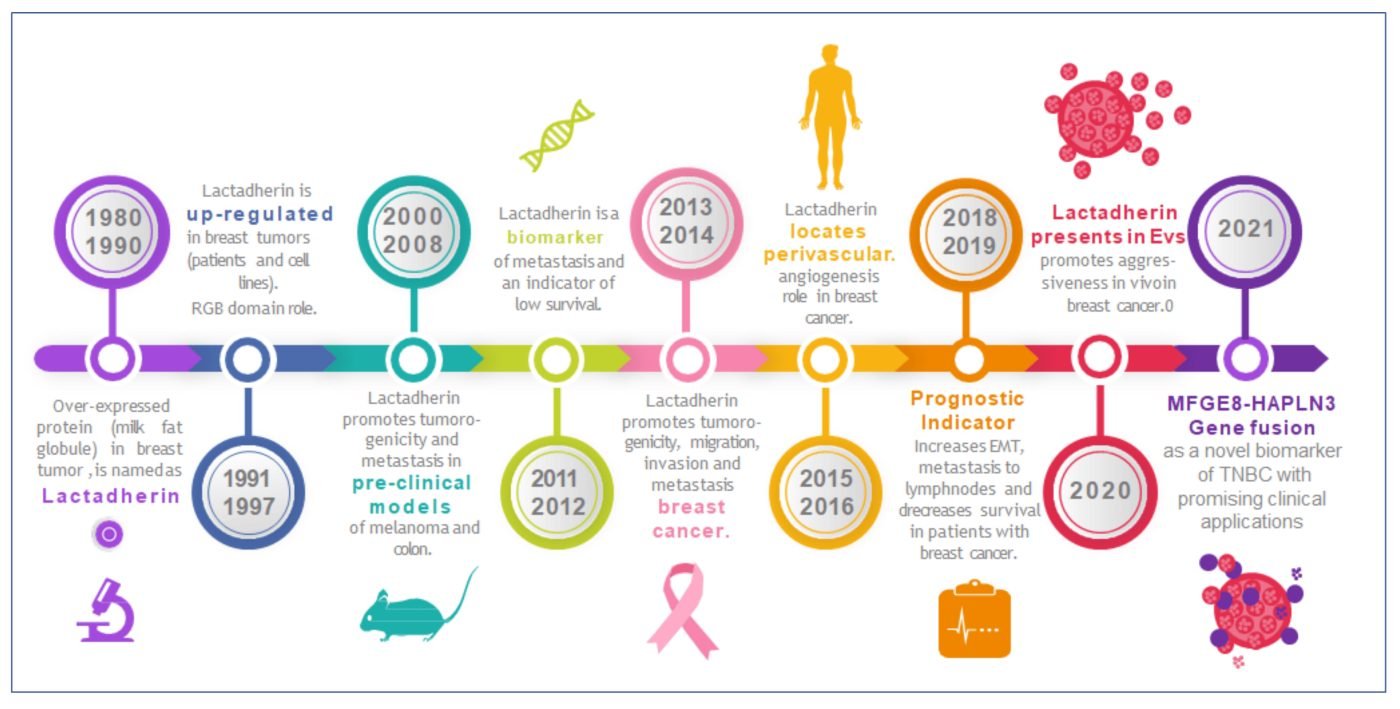
Figure 3 Timeline highlighting lactadherin research on its role in tumor development, progression, and metastasis. Lactadherin was first associated with breast tumors in the mid-80s to the early-90s. Since then, periods of fruitful research have been identified roles in tumor growth, progression, and metastasis. Lately, recent investigations have also identified lactadherin in EVs and exosomes, proposing it as a promising biomarker and/or therapeutic target. From: Durán-Jara E, Vera-Tobar T and Lobos-González L. (2022).