Biography
E-Mail: lleyton@uchile.cl
Dr. Lisette Leyton is a Biochemist from the Faculty of Chemical and Pharmaceutical Sciences, University of Chile and obtained her PhD in Sciences in the Faculty of Sciences, University of Chile. Dr. Leyton trained as a Post Doc at Duke University, Durham, NC, USA, and at University of Lausanne, Epalinges, Switzerland. She is currently a Professor of the Faculty of Medicine, University of Chile and also the Coordinator of the Committee of the Joint Degree Doctoral Program in Biomedical Sciences of the same Faculty.
Research Interest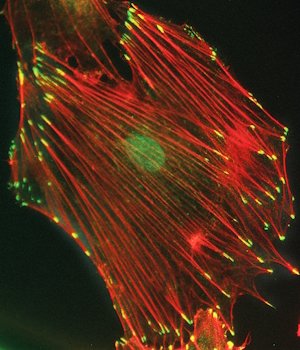
⮚ Signal Transduction Pathways:
- Cell-cell and cell-matrix interactions initiate signal transduction pathways.
- Rho GTPases play a role in cell adhesion and migration.
⮚ Neuron-Astrocyte Interaction:
- Neuronal Cell Death: Occurs under pro-inflammatory stimuli.
- Astrocyte Reactivity: Upon injury or inflammation, astrocytes become reactive. They migrate, increase in size, and participate in the formation of the glial scar.
- Glial Scar Formation: Generates a non-permissive environment for neuronal regeneration.
⮚ Molecular Mechanisms in astrocytes:
- Thy-1/CD90 Interaction with αvβ3 Integrin: Clusters αvβ3 integrin in astrocytes. Induces tyrosine phosphorylation of focal adhesion proteins. Activates RhoA and form focal adhesions and stress fibers.
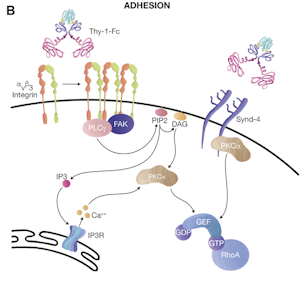
From: Valdivia A, Avalos AM and Leyton L (2023).
- Additional Interaction with Syndecan-4 in a ternary complex.
- FAK, PLCγ, PI3K, AKT, PKCα and RhoA-Dependent Pathways.
- ATP Release: Through Connexin43 and Pannexin1 hemichannels.
- Activation of P2X7 Channels: By increased ATP levels, permitting calcium entry into the cell.
⮚ Temporally Regulated Signaling:
- Prolonged stimulation with Thy-1/CD90 leads to disassembly-assembly of focal adhesions and astrocyte migration in a PI3K and Rac1-dependent manner.
⮚ β3 Integrin Overexpression:
- Triggered by activation of NFkB. αVβ3 integrin Induces a reactive phenotype that respons to Thy-1/CD90.
⮚ Potential Modulation of Astrocyte Reactivity by controlling β3 Integrin levels.
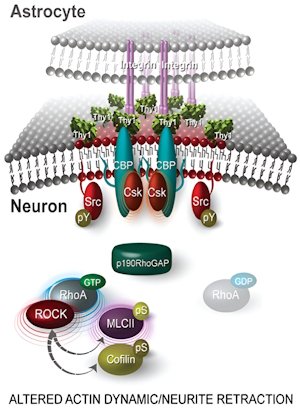
De: Maldonado et al 2017.
⮚ αVβ3 Integrin as Ligand for neuronal Thy-1/CD90. Upon binding, restricts neurite growth and induces retraction of existing processes.
⮚ Molecular Mechanisms in neurons:
- Thy-1/CD90 Clustering induced upon interaction with αVβ3 integrin. Recruitment and inhibition of Src. Downstream pathways involve CBP and Csk, p190RhoGAP/RhoA/ROCK axis. MLCII and Cofilin.
- Responses include Neurite Contraction.
Therefore, Thy-1/CD90 has a bimodal function: it acts as a receptor on neuronal cells; serves as a ligand for αvβ3 integrin on astrocytes. Implications for Astrogliosis, a process triggered in astrocytes after brain injury that precludes neuronal regeneration and repair.
Expected Insights: Better understanding of molecular mechanisms controlling Neurite outgrowth and Astrocyte function.
Selected References
- Leyton, L., Schneider, P., Labra, C.V., Rüegg, C., Hetz, C.A., Quest, A.F.G. and C. Bron. “Thy-1 binds to integrin ß3 on astrocytes and triggers formation of focal contact sites”. Current Biology. 11:1028-38 (2001). PubMed Link
- Ávalos, A.M., Labra, C., Quest, A.F.G., and Leyton, L. “Signaling triggered by Thy-1 interaction with ß3 integrin on astrocytes is an essential step towards unraveling neuronal Thy-1 function”. Biol. Res. 35: 231-238 (2002). PubMed Link
- Avalos AM, Arthur WT, Schneider P, Quest AF, Burridge K, Leyton, L. “Aggregation of integrins and RhoA activation are required for Thy-1-induced morphological changes in astrocytes”. J Biol Chem. Sep 10;279(37):39139-45. Epub 2004 Jun 25 (2004) Abstract. PubMed Link
- Hermosilla, T., Muñoz, D, Herrera-Molina, R., Valdivia, A., Nham, S.-U., Schneider, P., Burridge, K., Quest, A.F.G., Leyton, L. “Direct Thy-1/integrin αvβ3 interaction mediates neuron to astrocyte communication”. BBA Mol Cell Biol, 1783: 1111-1120 (2008). PubMed Link
- Avalos, A.M., Valdivia, A.D., Muñoz, N., Herrera-Molina, R., Tapia, J.C., Lavandero, S., Chiong, M., Burridge, K., Schneider, P., Quest, A.F.G., and Leyton, L. “Neuronal Thy-1 induces astrocyte adhesion by engaging syndecan-4 in a cooperative interaction with αvβ3 integrin that activates PKCα and RhoA”. J. Cell Sci. 122:3462-3471 (2009). PubMed Link
- Henríquez, M, Herrera-Molina, R., Valdivia, A., Alvarez, A., Kong, M., Muñoz, N., Eisner, V., Jaimovich, E., Schneider, P., Quest, A.F.G.*, Leyton, L.* “ATP release due to Thy-1-integrin binding induces P2X7-mediated calcium entry required for focal adhesion formation”. J. Cell Sci.124: 1581-1588 (2011) *equal contribution as corresponding authors. PubMed Link
- Herrera-Molina, R., Frischknecht, R., Maldonado, H., Seidenbecher, C.I., Gundelfinger, E.D., Hetz, C., Aylwin, M.L., Quest, A.F.G., and Leyton, L. “Astrocytic αVβ3 Integrin Inhibits Neurite Outgrowth and Promotes Retraction of Neuronal Processes by Clustering Thy-1”. PLoS One. 7(3):e34295. Epub 2012 Mar 30 (2012). PubMed Link
- Kong, M., Muñoz, N., Valdivia, A., Alvarez, A., Herrera-Molina, R., Cárdenas, A., Schneider, P., Burridge, K., Quest, A.F.G., and Leyton, L. “Thy-1-mediated Cell -Cell Contact Induces Astrocyte Migration through the Engagement of αVβ3 Integrin and Syndecan-4”. BBA-Mol Cell Res. 1833:1409-1420 (2013). PubMed Link
- Alvarez, A., Lagos-Cabré, R., Kong, M., Cárdenas, A., Burgos-Bravo, F., Schneider, P., Quest, A.F.G., Leyton, L. “Integrin-mediated transactivation of P2X7R via hemichannel-dependent ATP release stimulates astrocyte migration”. BBA-Mol Cell Res. 1863: 2175–2188 (2016). PubMed Link
- Maldonado, H., Calderon, C., Burgos-Bravo, F., Kobler, O., Zuschratter, W., Ramirez, O., Härtel, S., Schneider, P., Quest, A.F.G., Herrera-Molina, R.*, Leyton, L.* “Astrocyte-to-neuron communication through integrin-engaged Thy-1/CBP/Csk/Src complex triggers neurite retraction via the RhoA/ROCK pathway”. BBA-Mol Cell Res. 1864:243-254 (2016). *Corresponding authors (equal contribution). PubMed Link
- Lagos-Cabré, R., Alvarez, A., Kong, M., Burgos-Bravo, F., Cárdenas, A., Rojas-Mancilla, Pérez-Nuñez, R., Herrera-Molina, R., Rojas, F., Schneider, P., Herrera-Marschitz, M., Quest, A.F.G., van Zundert, B., and Leyton, L. “aVb3 Integrin regulates astrocyte reactivity”. J. Neuroinflammation 14:194 (2017). doi: 10.1186/s12974-017-0968-5. PubMed Link
- Lagos-Cabré, R., Brenet, M., Díaz, J., Pérez, R. D., Pérez, L. A., Herrera-Molina, R., Quest, A.F.G., and Leyton, L. “Intracellular Ca2+ increases and Connexin 43 hemichannel opening are necessary but not sufficient for Thy-1-induced astrocyte migration”. Int. J. Mol. Sci. 19, 2179–2195 (2018). doi: 10.3390/ijms19082179. PubMed Link
- Leyton, L., Díaz, J., Martínez, S., Palacios, E., Pérez, L.A. and Pérez, R.D. “Thy-1/CD90 a Bidirectional and Lateral Signaling Scaffold”. Front. Cell Dev. Biol. 7:132 (2019). doi: 10.3389/fcell.2019.00132. PubMed Link
- Valdivia, A., Cárdenas, A., Brenet, M., Maldonado, H., Kong. M., Díaz, J., Burridge, K., Schneider, P., San Martín, A., García-Mata, R., Quest, A.F.G., Leyton, L. “Syndecan-4/PAR-3 signaling regulates focal adhesion dynamics in mesenchymal cells”. Cell Communication and Signaling. 2020 Aug 18;18(1):129. PubMed Link – Open Access Link – News Link
- Burgos-Bravo, F., Martínez-Meza, S., Quest, A.F.G., Wilson, C.A.M. and Leyton, L. “Application of Force to a Syndecan-4 Containing Complex With Thy-1–αVβ3 Integrin Accelerates Neurite Retraction”. Front. Mol. Biosci., 29 September 2020. Open Access Link
- Pérez, L.A., Rashid, A., Combs, J.D., Schneider, P., Rodríguez, A., Salaita, K. and Leyton, L. “An Outside-In Switch in Integrin Signaling Caused by Chemical and Mechanical Signals in Reactive Astrocytes”. Front. Cell Dev. Biol. 2021 Aug 23;9:712627. PubMed Link – Open Access Link
- Pérez, L.A., Leyton, L., Valdivia, A. “Thy-1 (CD90), Integrins and Syndecan 4 are Key Regulators of Skin Wound Healing”. Front. Cell Dev. Biol. 2022 Feb 03;10:810474. PubMed Link – Open Access Link
- Pérez-Núñez, R., Chamorro, A., González, M.F., Contreras, P, Artigas, R., Corvalán, A.H., van Zundert, B., Reyes, C., Moya, P.R., Avalos, A.M., Schneider, P., Quest, A.F.G. & Leyton, L. “Protein kinase B (AKT) upregulation and Thy-1-αvβ3 integrin-induced phosphorylation of Connexin43 by activated AKT in astrogliosis”. J Neuroinflammation. 2023 Jan 6;20(1):5. PubMed Link – Open Access Link – Springer Link
- Palacios, E., Lobos-González, L., Guerrero, S., Kogan, M.J., Shao, B., Heinecke, J.W., Quest, A.F.G., Leyton, L., Valenzuela-Valderrama, M. “Helicobacter pylori outer membrane vesicles induce astrocyte reactivity through nuclear factor-κappa B activation and cause neuronal damage in vivo in a murine model”. J Neuroinflammation. 2023 Mar 9;20(1):66. PubMed Link – Open Access Link – Springer Link


